 What would you see when rubidium is dropped into water? Slow reaction with cold water, vigorous with hot water Reactivity series of metals Order of reactivity Which metal will react readily with cold water? This is because according to the periodic properties the reactivity of potassium and chlorine is very high. Now among the given options, the reaction between Chlorine and potassium will be most violent. Read More: What are 3 sources of energy for metabolism? Which pair of elements will react together most violently? Sodium does not react with nitrogen, so sodium is usually kept immersed in a nitrogen atmosphere (or in inert liquids such as kerosene or naphtha). Which metal will not react with nitrogen? So, it reacts with fluorine, F 2, chlorine, Cl 2, bromine, I 2, and iodine, I 2, to form respectively rubidium(I) bromide, RbF, rubidium(I) chloride, RbCl, rubidium(I) bromide, RbBr, and rubidium(I) iodide, RbI. Rubidium metal reacts vigorously with all the halogens to form rubidium halides. Rubidium (Rb) has an atomic number of 37. … Rubidium is the first alkali metal in the group to have a density higher than water, so it sinks, unlike the metals above it in the group. Rubidium is a very soft, silvery-white metal in the alkali metal group. Rubidium is the chemical element with the symbol Rb and atomic number 37. Since it is easily ionized, it might be used as a propellant in ion engines on spacecraft. It is also used in the manufacture of photocells and in special glasses. Rubidium is used in vacuum tubes as a getter, a material that combines with and removes trace gases from vacuum tubes. 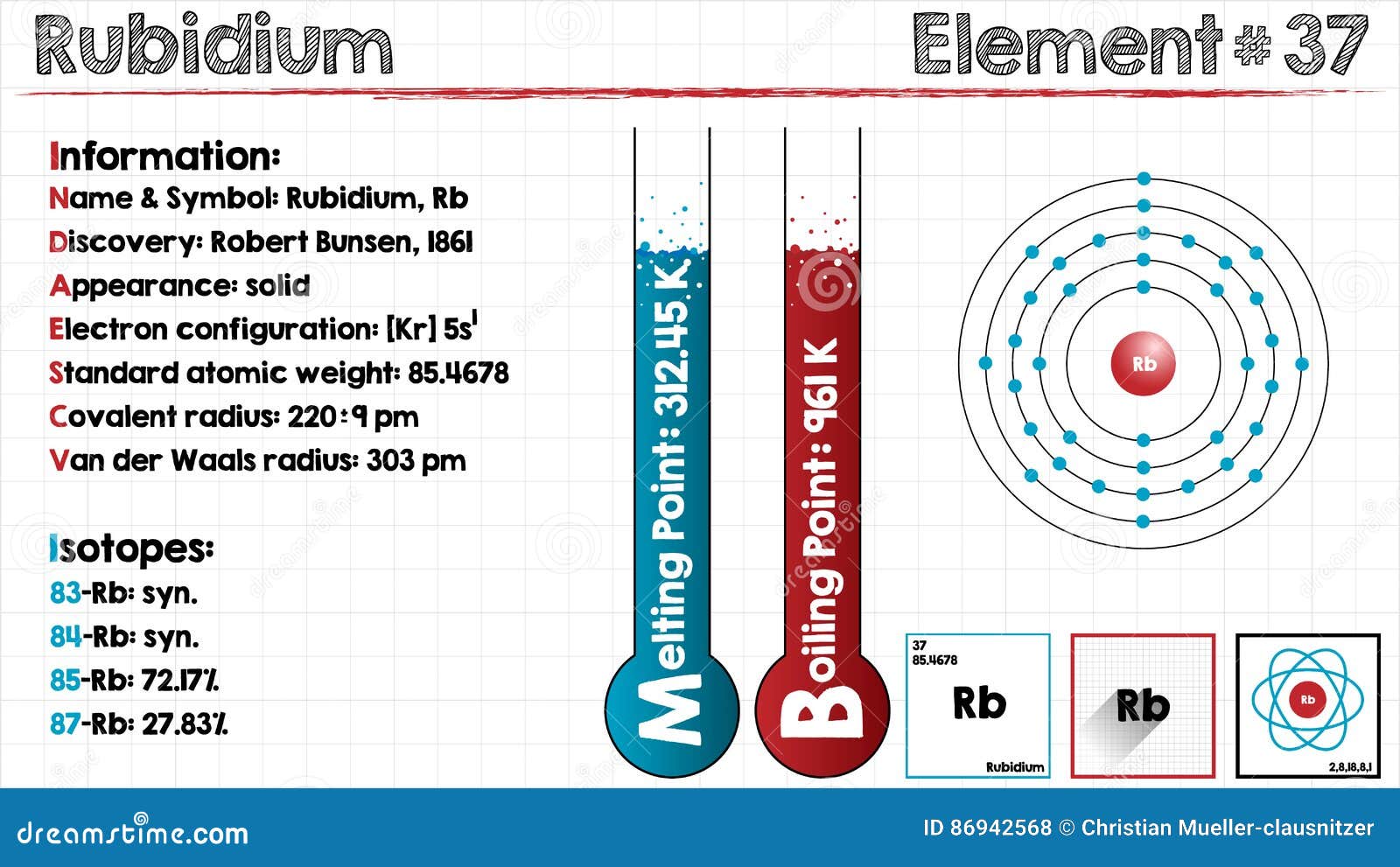
The chlorides are all white solids at room temperature, and dissolve in water to make a neutral solution. The group 1 metals all react with chlorine to produce chlorides. Namesĭoes chlorine and rubidium form an ionic compound? How do alkali metals react with chlorine? Rubidium hydroxide is formed when rubidium metal reacts with water. Read More: What are flash memory cards used for? Is rubidium soluble in water? The heavier alkali metals will react more violently with water. The heat produced by this reaction may ignite the hydrogen or the metal itself, resulting in a fire or an explosion. Why do the alkali metals react with water?Īlkali metals react with water to produce heat, hydrogen gas, and the corresponding metal hydroxide. … Rubidium forms alloys with other alkali metals as well as with elements such as mercury (known as amalgams) and gold, according to New World Encyclopedia. They have one electron in their outer shell, and do not occur freely in nature, according to ChemicalElements. Why is rubidium so reactive?Īlkali metals are highly reactive. All of these metals react vigorously or even explosively with cold water. It uses these reactions to explore the trend in reactivity in Group 1. This page looks at the reactions of the Group 1 elements – lithium, sodium, potassium, rubidium and caesium – with water. Question: Rubidium and nitrogen react in a combination reaction to produce rubidium nitride: 6 Rb(s) + N2(g) 2 Rb3N(s) How many moles of rubidium are needed to produce 3.00 mol of Rb3N when the reaction is carried out in the presence of excess nitrogen. Also, this metal reacts very violently in the presence of air and water. It is the second most electropositive element on the periodic table, so it very easily gives up its one electron to form ionic bonds. Rubidium is a member of the group 1 elements known as alkali metals. So, it burns with chlorine, Cl 2, to form rubidium(I) chloride, RbCl. While not a normal route of preparation because of the expense, rubidium metal reacts vigorously with all the halogens to form rubidium halides. Rubidium hydroxide solution and hydrogen are formed. It reacts violently and immediately, with everything leaving the container. Rubidium: Rubidium sinks because it is less dense than water. Rubidium is a solid at standard temperature and pressure with a melting point of 39.3 ☌.Reaction of rubidium with bases Rubidium does not react with bases, under normal conditions, but reacts with the water instead. Rubidium reacts explosively with water to produce Hydrogen gas and Rubidium Hydroxide. Rubidium oxidises extremely quickly in the presence of Oxygen so it must be stored in a completely sealed container. Rubidium is more reactive than Carbon on the reactivity series so it must be extracted from its ore using electrolysis. Properties Rubidium is a more reactive alkali metal than Potassium but less reactive than Caesium. Rubidium ions have lost an electron to become positively charged. An atom of Rubidium has only 1 electron in its outer shell. Atomic Structure The most stable isotope of Rubidium has 48 neutrons in its nucleus giving it an atomic mass of 85. Rubidium atoms join together in a giant metallic structure. 
Rubidium is a Group 1 element, on the Periodic Table, with 37 protons in the nucleus.Ībout Rubidium Molecular Structure Rubidium has the chemical symbol Rb. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
